Understand What’s Required Without the Guesswork
17″ – 19″ Transfer Height
Is the Enforceable Standard
The U.S. Department of Justice (DOJ) and Department of Health & Human Services (HHS) have established a 17″ – 19″ patient transfer height range as the enforceable standard for accessible exam and procedure tables.
Only 10 – 20% of Tables
Must Comply
Facilities are not required to replace every exam or procedure table. Current guidance requires that 10–20% of exam tables be accessible, depending on the size and type of the healthcare facility.
Compliance Deadlines
Are Approaching
Implementation timelines are already underway. Federally funded providers must comply by July 8, 2026, while state and local government facilities must comply by August 9, 2026. Now is the time to begin planning for compliance.
Federal Compliance Deadline
State and Local Compliance Deadline
Days
Days
Accessible Solutions Designed to Meet
DOJ and HHS ADA Standards
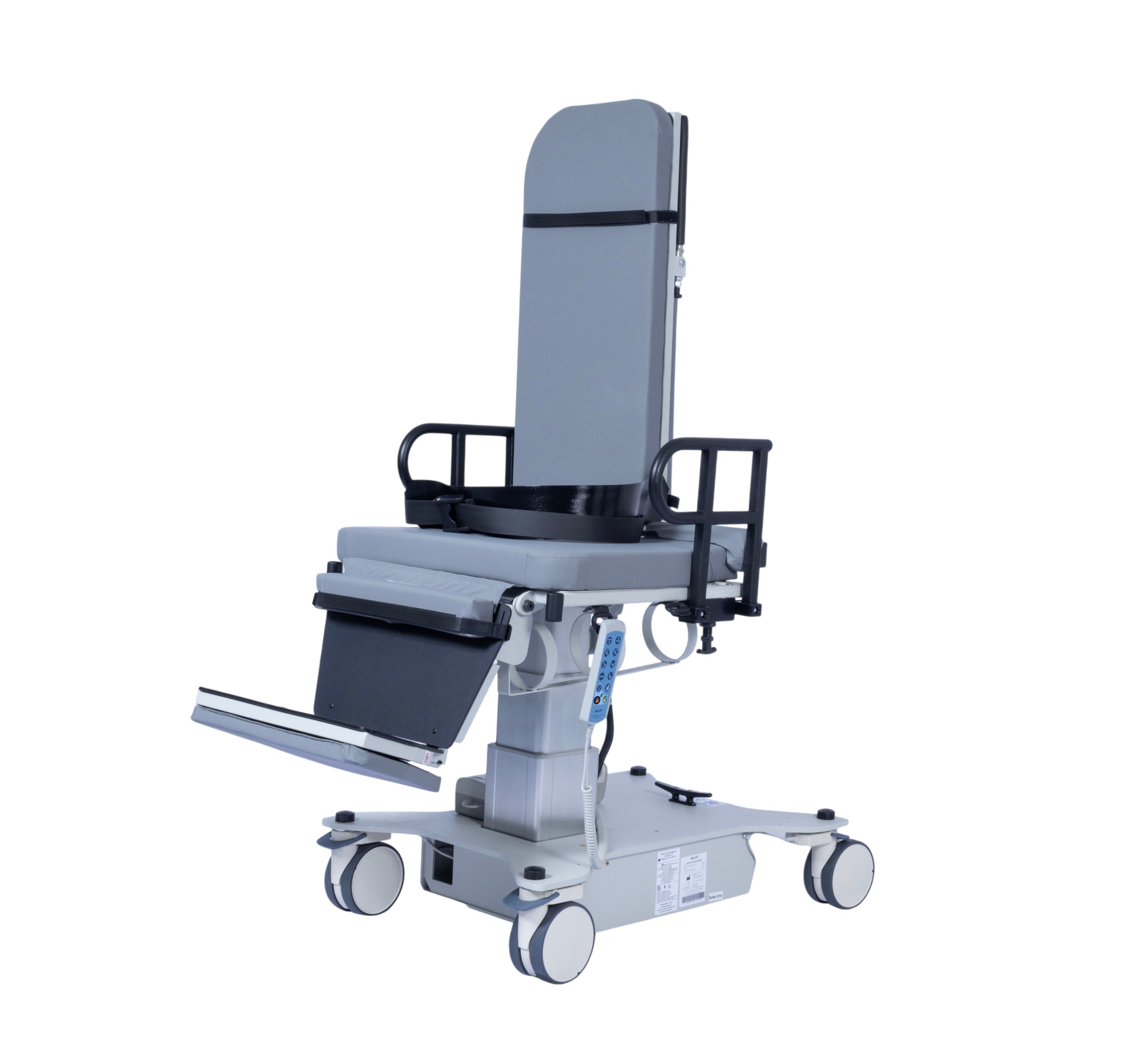
Introducing EchoBed®
IMPROVED TARGET FEATURES
- ADA 19″ AND U.S. ACCESS BOARD 17″ ENTRY HEIGHT
- INCREASED PATIENT WEIGHT CAPACITY OF 750 LBS.
- LARGE ELECTRIC PATIENT BACK SUPPORT
- ELECTRIC LOCKING CASTERS
- DEEPER IMAGING DROP SECTION OPENING
- FLEXIBLE AND ACCESSIBLE HAND CONTROL HOOKS
EchoBed® is under development and not yet commercially available. Specifications, features, and availability are subject to change pending final design certification.
FAQs
What is the current enforceable regulation for exam tables?
As of 2024, the DOJ and HHS enforce the 2017 U.S. Access Board standards, requiring exam tables to have a low transfer height between 17″ – 19″.
Do all exam tables need to meet the 17″ – 19″ standard?
No. Facilities must ensure that 10–20% of their exam tables (and other diagnostic equipment) meet the standard, depending on the healthcare setting.
What about the newer 17″ height recommendation I’ve heard about?
In 2024, the U.S. Access Board issued a new recommendation to lower the minimum transfer height to 17″.
This is not enforceable. For it to become law, DOJ and HHS would need to initiate a formal rulemaking process—a lengthy, uncertain step that hasn’t been started.
What is the difference between a recommendation and a regulation?
A recommendation is a proposed best practice from the Access Board. A regulation becomes enforceable only when adopted by DOJ or HHS through rulemaking. Currently, the enforceable regulation is the 17″ – 19″ range.
Who Must Comply?
Federally Funded Clinics and Facilities
Enforced under Section 504 of the Rehabilitation Act by HHS (Health and Human Services).
Applies to providers that receive federal financial assistance (e.g., Medicare, Medicaid).
State and Local Government Healthcare Facilities
Enforced under Title II of the ADA by the DOJ (Department of Justice).
Applies to public hospitals, clinics, and any healthcare setting operated by state/local governments.
Does the regulation apply only to exam tables?
No. The standards also cover scales, diagnostic chairs, and imaging equipment—not just exam tables. Requirements vary by equipment type.
Are any of MedicalPositioning products compliant?
Yes. The EchoBed®, VasScan Table™ X, and the MultiView® are ADA compliant.
When do providers need to be compliant?
July 8, 2026 – Federally funded facilities must have at least one compliant exam table and scale.
August 9, 2026 – State and local facilities must meet the same requirement.
What other accessibility features are required beyond transfer height?
Transfer surface size and firmness
Lift compatibility
Support rails or assist handles
Wheelchair accessibility
Reachable controls
Standing support and communication features
Additional Resources
REPERTOIRE MAGAZINE – MDE RULE SUMMARY
A distributor-focused overview that simplifies the impact of the new regulations.
https://repertoiremag.com/new-requirements-for-medical-diagnostic-equipment.html
FEDERAL REGISTER – DOJ RULE ON MDE ACCESSIBILITY
Official publication detailing the DOJ’s rule on the accessibility of medical diagnostic equipment for state and local governments.
https://www.federalregister.gov/documents/2024/08/09/2024-16889/nondiscrimination-on-the-basis-of-disability-accessibility-of-medical-diagnostic-equipment-of-state
EPSTEIN BECKER GREEN LAW FIRM – ADA COMPLIANCE FOR MDE WEBINAR
Hosted by legal experts at Epstein Becker Green, this insightful session breaks down the new regulations, clears up industry confusion, and answers key questions about what healthcare providers and suppliers need to do to comply. (Registration required to access.)
https://www.ebglaw.com/insights/events/breaking-barriers-toward-inclusive-health-care-new-ada-title-ii-regulations-require-accessible-medical-diagnostic-equipment
ADA NATIONAL NETWORK – OVERVIEW OF MEDICAL EQUIPMENT ACCESSIBILITY
Useful ADA guide for understanding equipment accessibility in healthcare settings.
https://adata.org/factsheet/accessible-medical-examination-tables-and-chairs
GATHERING STRENGTH – OVERVIEW OF NEW MDE RULES
Provides an overview of the new rules on medical diagnostic equipment and their impact on healthcare access.
https://gatheringstrength.org/new-rules-on-medical-diagnostic-equipment-improve-healthcare-access/